Science Topics – 121
Mitochondrial DNA (mt-DNA) encodes 22 species of tRNA (mt-tRNA), which contains unique posttranscriptional modifications. In animals, five mt-tRNAs contains taurine-modification at position 34U of the anticodon. A unique feature of the taurine-modifiation is its relevance with mitochondrial disease including mitochondrial encephalopathy, lactic acidosis, stroke-like syndrome (MELAS), and myoclonus epilepsy with ragged-red fibers (MERRF). It has been reported that taurine-modification was decreased in MELAS and MERRF patients, but the molecular function remained unclear. In the present study, we showed that taurine-modification is catalyzed by mitochondrial optimization 1 (Mto1) in mammals. Mto1 deficiency severely impaired mitochondrial translation and respiratory activity. Furthermore, the catastrophic mitochondrial dysfunction impairs trafficking of nuclear DNA-encoded mitochondrial proteins into inner membrane. The mistargeted proteins were aggregated and misfolded in the cytoplasm, which induced cytotoxic unfolded protein responses and amplified mitochondrial dysfunction. Importantly, application of chemical chaperons successfully suppresses cytotoxicity by reducing protein misfolding and increasing functional mitochondrial proteins in Mto1-deficient cells and mice. Thus, our results demonstrate the essential role of taurine-modification in mitochondrial translation and reveal an intrinsic protein homeostasis network between the mitochondria and cytosol, which has a therapeutic potential for mitochondrial diseases.
Md Fakruddin, Fan-Yan Wei, Takeo Suzuki, Kana Asano, Takashi Kaieda, Akiko Omori, Ryoma Izumi, Atsushi Fujimura, Taku Kaitsuka, Keishi Miyata, Kimi Araki, Yuichi Oike, Luca Scorrano, Tsutomu Suzuki, and Kazuhito Tomizawa
Cell Reports. vol. 22. 482-496, 2018
http://dx.doi.org/10.1016/j.celrep.2017.12.051
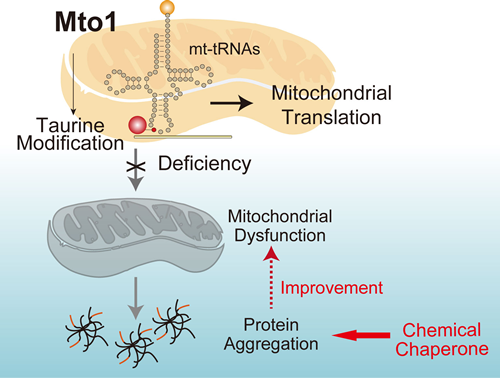
<Figure Legends>
Taurine-modification of mitochondrial tRNA is essential for protein translation in mitochondria. Deficiency of taurine-modification impaires mitochondrial translation, which induces defective mitochondrial respiration and abnormal membrane integrity. The severe mitochondrial dysfunction impairs the trafficking of mitochondrial proteins, which are subsequently misfolded and accumulated in cytosol and triggers cytotoxic unfolded protein response. Application of chemical chaperones prevents protein aggregation and suppress cytotoxicity in cells and mice having defective taurine-modification.
Department of Molecular Physiology, Faculty of Life Sciences, Kumamoto University, Kumamoto 860-8556, Japan
